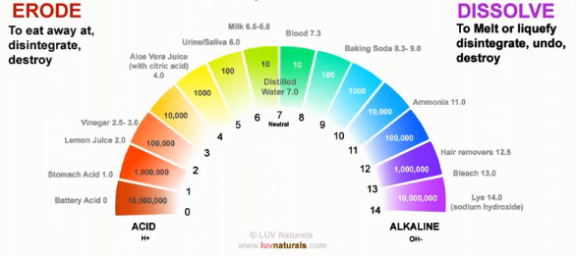
In the previous article, we covered the basics of pH balance. We know that the pH of hair is between 4.5 and 5.5. In this article, I will talk about how we sometimes, unknowingly, take the pH of our hair on a roller coaster ride.
What is the Ideal pH Range For Our Hair?
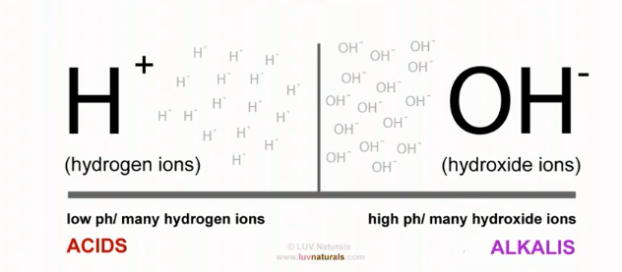
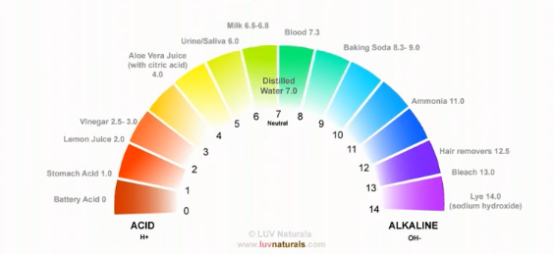
We already know that the pH range of hair is 4.5 to 5.5. This pH is based on the natural oil we secrete from our scalp (sebum). Hair and skin have the same pH.
What is in Sebum?
This oil on the surface of our skins is a complex mixture of fatty acids, glycerides, wax esters and cholesterols, etc., that we produce internally.
Sebum has the following functions:
- It reduces water loss from the skin surface
- It protects the skin from infections by unwanted bacteria and fungi
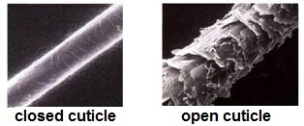
The Ideal pH range to keep our hair and skin in is between 4.5 – 5.5, this acidic pH helps to close our cuticles, hence holding moisture in our hair. It also wards off unwanted bacteria and fungi on our scalp.
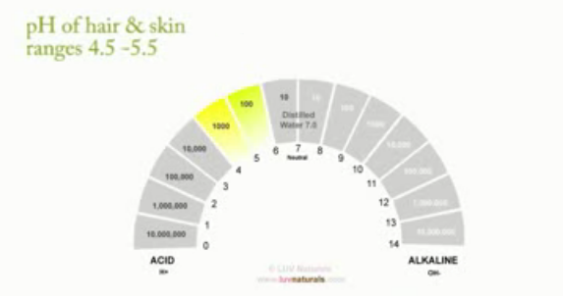
When alkaline solutions are placed on our hair and scalp, the exact opposite happens!
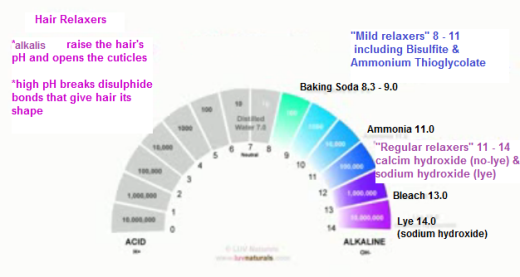
The cuticle layers are lifted. In the case of hair relaxers or hair dye, it is necessary for the cuticle layer to be open in order to change the protein structure of the hair.
For Afro textured hair, a relaxer with a pH of 11 or higher is usually required to make our hair permanently straightened.
This alkali pH lifts the cuticle layers wide open and also breaks and then reforms some of the disulphide bonds in our hair (disulphide bonds give our hair its shape). This process can be damaging, especially when done incorrectly.
After the hair is disrupted, it is neutralized with a very acidic solution to close the cuticle once again and stop the alkali chemicals from working.
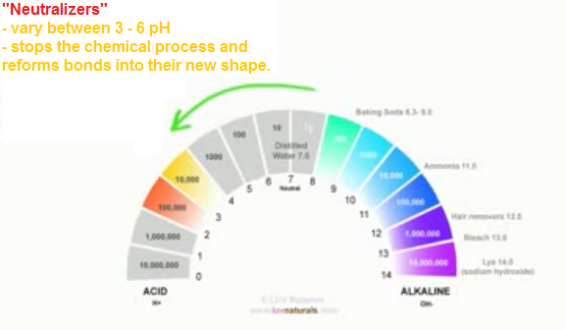
This is a roller coaster ride for our hair. The pH is taken from 4.5 up to 8.0+ and then back again, usually within minutes. Bringing the hair back to its acidic pH doesn’t change the fact that you’ve disturbed the hair in the first place.
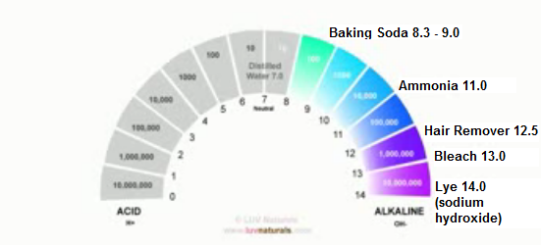
The baking Soda and Apple Cider Vinegar Method:
Some people have sworn by the use of baking soda as a natural method for washing our hair because they think it is better than shampoo. Kim Love explains how this is far from the truth using the following scientific facts and NOT OPINION:
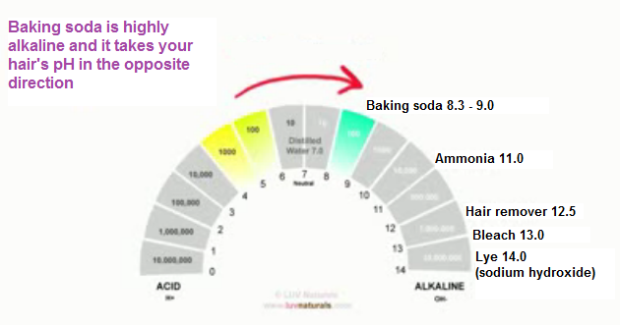
1. Baking soda, which has a pH of 8.3 or higher (even when diluted with water), is more alkaline than our hair. Like all alkalis, it takes our hair and scalp in the opposite direction of the ideal pH range.
2. This alkaline nature lifts our cuticle layers and has the potential to temporarily disrupt the bonds of our hair. People who use baking soda to wash their hair often say it gives them a looser curl pattern and softens the the hair. That is what an alkaline does. By disrupting the bonds in our hair, alkalis make the hair more unrestricted and ready for any new reshaping, hence the softness.
3. After rinsing with baking soda, to stop the chemical reaction, an apple cider vinegar rinse is used to bring the hair back to an acidic range. 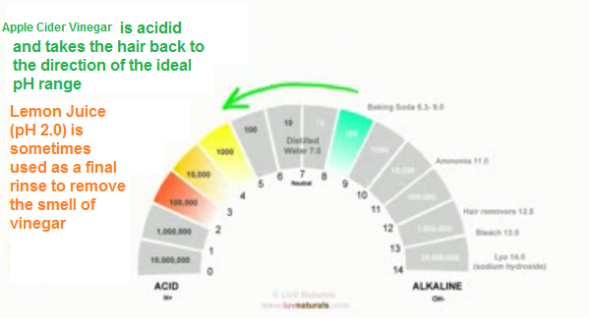
Similar to relaxers, this does not undo the disturbance done to the hair and scalp. Our scalps are naturally acidic to prevent bacteria and fungi from infecting the skin. Lots of dandruff problems and scalp conditions are aggravated and sometimes caused by high alkaline solutions.
Usually, when the apple cider vinegar is introduced, a burning sensation is felt. Even though it burns, the cuticles have opened up and the hair must now it must be neutralized to close the cuticles and re-form those hair bonds. Sound familiar?
In the next and FINAL article, I will explain how pH balancing can be applied and used properly in your hair care regimen.
Source: http://www.youtube.com/playlist?list=PL5532B6CD987E1D88