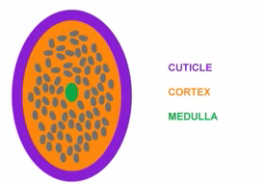
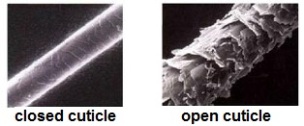
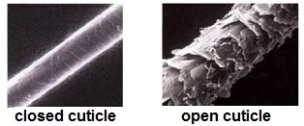
In the last article, I covered the basic structure of hair and explained what part of hair helps in retaining moisture. This part of hair is called the cuticle and it has an overlapping tree like structure that can either be open or closed. Take a look at the image below.
I left you guys asking, “just how do I encourage my cuticles to lay down or close?” and this article answers your questions. After lots of research, I came across a video series by youtube natural hair guru, Kim Love! (Kim, thanks so much for this multitude of information). If you ever have the time, check out her series on the structure of hair: http://www.youtube.com/playlist?list=PL5532B6CD987E1D88. You will not be disappointed.
So, I know of 3 different forces that the cuticle responds to:
- Temperature: Heat causes the cuticle layers to expand/lift open, whilst the cold causes them to contract/close. This is why frequently blown out or straightened hair, ends up damaged and in the winter our hair breaks because our cuticle layers are closed shut, not allowing moisture to enter the hair shaft.
- pH balance: Alkaline solutions cause the cuticle layers to open, while more acidic solutions causes them to close.
- Surface treatment: Running your hands downwards closes the cuticle layers. Also, sticky products like silicons or oils can manually close the cuticle’s layers. CAUTION! Even though silicons close the cuticle’s layers, they are not good for the hair. Yes, they seal moisture in the hair, BUT they can also keep well needed moisture out.
Of the 3 methods, pH balance is the most effective and yet least harmful to the hair, WHEN USED CORRECTLY.
Basics of pH Balance
In Kim’s basics of pH balance video, she tests the pH of different solutions using pH testing strips. Note that, water has a neutral pH of 7. The 3 solutions tested were vinegar, apple cider vinegar and baking soda. Both types of vinegars are known to be acidic solutions, whilst baking soda is known to be an alkali solution.
The test results revealed that:
- Vinegar has a pH that falls somewhere between 2 and 3. Which is an accurate result because vinegar has a well-known pH of about 2.5.
- Now, you may think that all vinegars are the same, but apple cider vinegar is slightly less acidic and has a pH of 3.
- The baking soda was dissolved in a bit of water to make a solution (you can only test the pH of liquids) and when tested, it revealed a pH of 9. Baking soda is a well-known alkali, it usually has a pH between 8.3 and 9.
Hey, what happens when you mix an acid with an alkali? You create a solution with a neutral pH of 7. Confused? Don’t worry, I’m about to explain the science behind it.
What does pH mean? It means potential of Hydrogen or power of Hydrogen. The scale used for measuring the potential of hydrogen ranges from 0 to 14; 0 being the most acidic, 14 being the most alkali and 7 being neutral.

As I said earlier, when an acid and an alkali mix they make a solution that has a pH of 7. How exactly does this work?
- Vinegar, an acid, contains a lot of hydrogen ions, charged and eager to attach to something. Baking soda, an alkali, contains many hydroxide ions (hydroxide is made up of an oxygen molecule attached to a hydrogen molecule).
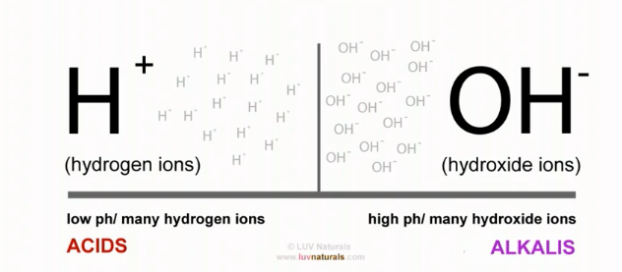
When you mix an acid with an alkaline, you take these substances from their extremes and bring them together, ending up with two of hydrogen and one of oxygen. H20?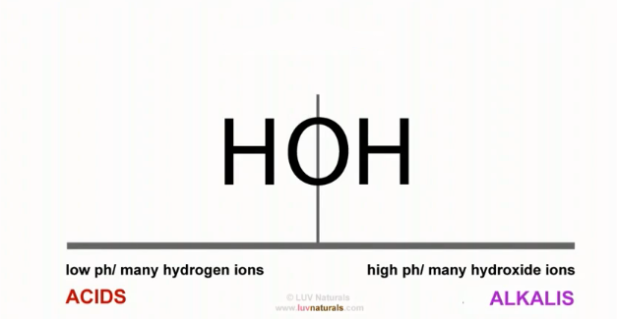

- So basically, you form water when the hydrogen and hydroxide connect. In essence, you neutralise them.
How does pH balance affect your everyday life?
- Take a look at this colourful scale below, representing the pH balance range. As you can see, battery acid is one of the strongest acids in our environment.
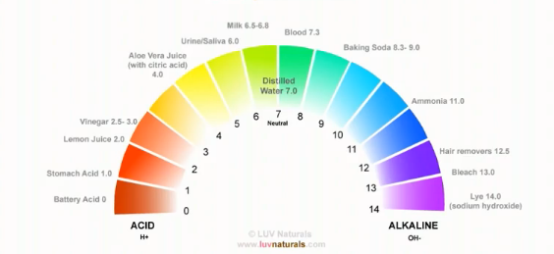
- Sodium hydroxide, also known as lye, used in hair relaxers, is one of the strongest alkali.
- Both extremes destroy almost anything in their path (I will elaborate on this point later).
- In the mean time, please take a moment to look over the scale. Notice where some of your household items and bodily fluids are located.
Human hair has a pH range of 4.5 to 5.5. From Kim’s research, this range can refer to the natural oil (also known as sebum), which we produce from our scalp (in other words, sebum has a pH range between 4.5 and 5.5) OR when hair is dissolved, it has a pH range between 4.5 and 5.5. Notice how our hair is more acidic than water?
Please note that these colour bars are a little misleading. You may think that one step up is not a big deal. pH balance is actually measured on a logarithmic scale. So, for every step you must multiply the number by 10. In other words, sodium hydroxide is 10 million times more alkaline than water. Battery acid is 10 million more acidic than water.
Remember I said these extremes will destroy almost anything in their path? Here are the verbs commonly used to describe the way acids and alkali destroy things differently. Both extremes are harmful to your hair.
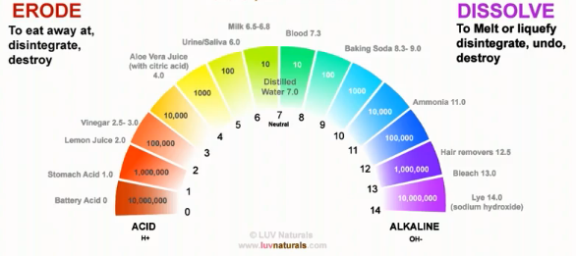
People in the natural hair community have experimented with baking soda and apple cider vinegar on their hair. The next article will explore some of the effects these products have on our hair and answer whether it makes sense to use them. Also, do you want to know what the ideal range of pH for hair is? Stay tuned for part 3!
Source:
- The Structure of Hair (2010), video, < http://www.youtube.com/playlist?list=PL5532B6CD987E1D88 > [07 July 2013].